- Western blotting technique is used for the separation and identification of a specific protein in a complex mixture of proteins based on their molecular weight.
- It is similar to Southern blotting (that detects DNA fragments) and Northern blotting (that detects mRNAs).
You may also want to see: Southern Blotting: Principle, Steps involved and Applications
Principle:
- In western blotting, an individual protein of interest is identified using radiolabelled or enzyme-linked polyclonal or monoclonal antibody specific for that protein.
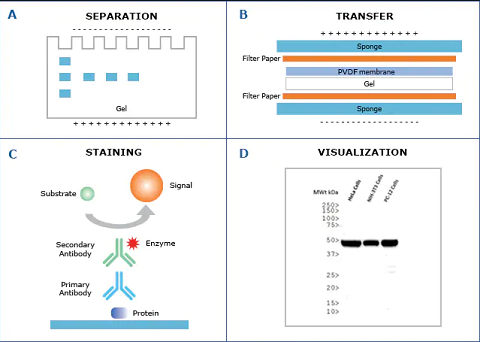
- A protein mixture is electrophoretically separated on an SDS-polyacrylamide gel (SDS-PAGE), a slab gel infused with a dissociating agent called Sodium dodecyl sulphate (SDS).
- The protein bands are transferred to a nitrocellulose membrane by an electrophoretic process called blotting where the proteins are then immobilized.
- The desired protein on the membrane can either be detected by the use of a labeled primary antibody directed against the protein or a labeled secondary antibody directed against the primary antibody in antigen-antibody complex.
- The antigen-antibody complexes can be visualized in a variety of ways;
- If the protein of interest is bound by radioactive antibody, its position on the blot is determined X-ray autoradiography.
- If enzyme-linked antibodies are employed, then addition of chromogenic substrate produces a highly colored and insoluble product at the site of target protein and makes it visible.
- A more sensitive way is the use of a chemiluminiscent compound with suitable enhancing agents to produce light at the protein site and make it visible.
Steps involved:
- Western blotting technique involves the following steps:
Step 1: Sample preparation or Cell lysis to extract protein:
- The most common form of sample for western blotting are the cell lysates.
- Proteins present in the cell cytosol are extracted by mechanical, chemical or enzymatic lysis of cell.
- This process is carried out at a lower temperature in the presence of protease inhibitor to prevent the denaturation of proteins.
- The appropriate concentration of protein in the sample is determined by a spectrophotometer.
- The sample is then diluted into a loading buffer, which contains glycerol that makes the sample sink easily into the wells of the gel.
- To monitor the movement of proteins in the gel slab, bromothymol blue (a tracking dye) is also added in sample before loading into the wells.
Step 2: Gel electrophoresis and separation of protein components
- Sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) is performed after loading the sample into the well.
- Proteins being negatively charged move towards the positive pole (anode) during electrophoresis.
- The protein components are separated according to their molecular weight, electric charge etc.
- Components with lower molecular weight migrate faster than the ones with higher molecular weight.
Step 3: Blotting or Electrotransfer:
- The gel is removed from the electrophoretic apparatus and applied to a protein-binding sheet of nitrocellulose or nylon or polyvinylidene fluoride (PVDF) membrane.
- The nitrocellulose membrane is placed or sandwiched between the gel and cassette of filter paper.
- The proteins in the gel are transferred to the nitrocellulose sheet by capillary action enhanced by the passage of the electric current. This process is called electro transfer or electro blotting.
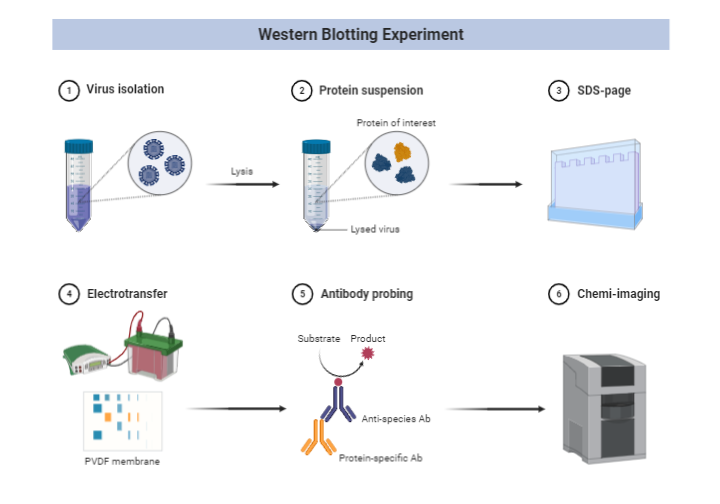
Image Credit:
https://app.biorender.com/biorender-templates/figures/5c95180bc2753f33003dacd3
Step 4: Blocking
- Nitrocellulose sheet is a protein-binding membrane and hence labeled antibodies being proteins are likely to bind the nitrocellulose membrane without forming antigen-antibody complex.
- This is the reason why the membrane is non-specifically saturated or masked by using casein or Bovine serum albumin (BSA) before adding the primary antibodies. This is called blocking.
- Casein or BSA blocks the labeled antibodies from binding to the nitrocellulose membrane.
Step 5: Flooding the membrane with radiolabeled Primary Antibody
- The primary antibody specific to the protein of interest is radiolabeled and added on the nitrocellulose membrane.
- This radiolabeled antibody forms Antigen-antibody complex with the desired protein.
Step 6: Treatment with enzyme-linked Secondary Antibody
- A second antibody called secondary antibody is linked to enzymes like alkaline phosphatase or Horseradish peroxidase (HRP) and added on the membrane already flooded with primary antibody.
- This secondary antibody is an antibody against primary antibody, hence also called anti-antibody. It binds to the primary antibody in the antigen-antibody complex.
Step 7: Treatment with suitable substrate to visualize the target protein
- The reaction mixture is incubated with a specific substrate.
- The enzyme converts the substrate to form an insoluble color product at the site of reaction.
- The position of the antibodies (or the target protein) is visualized as a colored band on the membrane.
- Alternatively, a chemiluminscent ELISA can be used to generate light that is readily detected by exposure of the blot to a piece of photographic film.
Applications:
- It can also identify a specific antibody in a mixture. In this case, known antigens of well-defined molecular weight are separated by SDS-PAGE and blotted onto nitrocellulose. The separated bands of known antigens are then probed with the sample suspected of containing antibody specific for one or more of these antigens.
- It helps in the determination of size and amount of protein in the given sample.
- The most widely used application of Western blotting is in the confirmatory testing for HIV. It determines whether the patient has antibodies against any viral proteins.
- It is useful in the detection of defective proteins in our body. E. g. Prions disease.
- It is applied as the definitive test for different diseases like Hepatitis B, Herpes, Lyme disease etc.
Western blotting: Principle, Steps involved and Applications
References:
- Kindt T. J, Goldsby R. A & Osbotne B. A, (sixth edition). Kuby Immunology, New York , NY: W.H. Freeman and Company, 2007
- https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3456489/#:~:text=Western%20blot%20is%20often%20used,a%20band%20for%20each%20protein.